The Shelley and Steven Einhorn Clinical Research Center
Established in 2010, The Shelley and Steven Einhorn Clinical Research Center at New York Eye and Ear Infirmary of Mount Sinai (NYEE) is the epicenter for research that’s helping to discover a new generation of treatments and diagnoses for degenerative eye diseases like diabetic retinopathy, glaucoma, macular degeneration, and much more. The Center provides a comprehensive setting for NYEE’s researchers and clinicians to conduct a growing portfolio of clinical investigations, often done in conjunction with pharmaceutical and other academic partners, all aimed at fostering the best patient care and life changing breakthroughs.
“Patients often come to New York Eye and Ear Infirmary because they know they can get the most up-to-date diagnostic facilities and access to bold new therapies they can’t find elsewhere,” says Richard Rosen, MD, Vice Chair and Director of Ophthalmology Research at NYEE.
Areas of Research
The scientists and physicians at the Einhorn Clinical Research Center work in eight distinct areas, each devoted to the development of better diagnostic technologies and clinical trials. The Center’s work targets specific eye diseases with the goals of preventing blindness and restoring vision. Areas of research include:
- Adaptive optics cellular imaging
- Optical coherence tomography clinical imaging
- Clinical trials and outcomes research
- Glaucoma
- Neuro-ophthalmology
- Ocular circulation, functional, and metabolic imaging
- Uveitis and ocular inflammation
- Retina diagnostics and restoration
State-of-the-Art Ocular Imaging Center
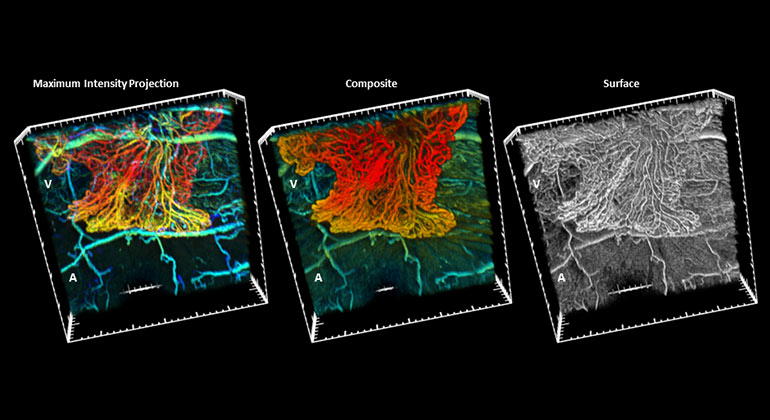
Advanced imaging is a critical component of the innovative care delivered at NYEE, supporting progress our researchers and clinicians are making in areas like stem cell and gene therapy, as well as the illumination of basic microanatomy at the cellular level. The Einhorn Center is at the forefront of developing and implementing a wide range of high resolution instruments to help diagnose and treat diseases of the eye. NYEE’s Advanced Retinal Imaging Laboratory is a unique facility exploring new technologies for imaging the human retina and understanding its anatomy, physiology, and visual function. As a leader in innovative structure and function studies of the eye, the Lab utilizes adaptive optics, optical coherence tomography and angiography, quantitative fluorescence, hyperspectral imaging, and microperimetry.
Advances in analytical software for adaptive optics images by researchers at Einhorn Center’s Marrus Adaptive Optics Imaging Laboratory are providing quantitative as well as qualitative assessment of the cellular structures directly involved in various retinal diseases.
“We’re committed to finding ways to detect diseases like macular degeneration, diabetic retinopathy, and glaucoma long before structural changes in the eye appear, so we can initiate treatment as soon as possible,” explains Dr. Rosen. “And thanks to the tireless work of our team of dedicated researchers at the Einhorn Center, we are actively advancing towards our goals.”
Clinical Trials
Since its inception, The Einhorn Clinical Research Center has hosted many exciting vision research studies, providing patients with access to diagnostic and therapeutic technologies years before they’re generally available. Drawing investigators from all eye subspecialties, the Center is focused on exploring the clinical benefits of a wide array of innovative medical and surgical techniques, therapeutics, and prevention programs, as well as expediting transfer of new laboratory discoveries in diagnostic methods and treatments to our patients. Numerous eye clinical trials are open to patients for enrollment.
Office of Research Services
Research conducted by Icahn School of Medicine at Mount Sinai faculty, at the New York Eye and Ear Infirmary of Mount Sinai, is subject to Mount Sinai’s ethical and legal requirements for the conduct and oversight of human research. The Office of Research Services (ORS) serves as a central resource for the Mount Sinai Health System research community, and oversight of human subjects research is provided by the Program for the Protection of Human Subjects (PPHS).
Research Faculty
Medical Leadership
James C. Tsai, MD, MBA
President - New York Eye and Ear Infirmary of Mount Sinai
Delafield-Rodgers Professor and Chair Department of Ophthalmology
Icahn School of Medicine at Mount Sinai
Paul A. Sidoti, MD
Chair, Department of Ophthalmology
New York Eye and Ear Infirmary of Mount Sinai
Chief, Glaucoma
Mount Sinai Health System
Professor of Ophthalmology
Icahn School of Medicine at Mount Sinai
Richard B. Rosen, MD, ScD (Hon), FACS, FASRS, CRA
Vice Chair and Director, Ophthalmology Research
Surgeon Director and Chief of Retina
New York Eye and Ear Infirmary of Mount Sinai
Professor of Ophthalmology
Icahn School of Medicine at Mount Sinai
Research Faculty
Rudrani Banik, MD
Toco Chui, PhD
Mar-Abigail Craven, MD
Sarita Dave, MD
Avnish Deobhakta, MD
Ronald Gentile, MD
Anita Gupta, MD
Meenakashi Gupta, MD
Gautam Kamthan, MD
Tsontcho A Ianchulev, MD
Gennady Landa, MD
Gareth M. C. Lema, MD, PhD
Kira Manusis, MD
Trisha Mor, MD
Harsha Reddy, MD
Richard B. Rosen, MD
Paul A. Sidoti, MD
Ted Smith, MD, PhD
Tak Yee Tania Tai, MD
Kateki Vinod, MD
Angie Wen, MD